at National Cancer Centre Singapore.
A Cancer Genetics Service (CGS) Consultation at National Cancer Centre Singapore.
From the vantage point of 2018, it is difficult to imagine a time when cancer was not widely accepted as a genetic disease, in the most basic sense of being caused by alterations in the structure and function of genes. Indeed, it was not until the second half of the 20th century that the heritable nature of common cancers started to be widely accepted. Over the decades since then, genealogic and epidemiologic observations set the stage for a burst of scientific activity in the 1990s, when the discovery of genes responsible for cancer predisposition syndromes was made.
These discoveries provided valuable insights to the biology of cancers, paved the way for integration of genetics in cancer risk assessment and realising the potential of gene-directed management and treatment.
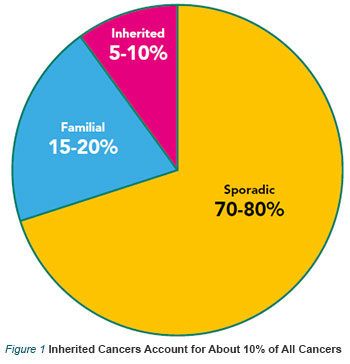
Currently, over 400 hereditary cancer susceptibility syndromes have been described, most of which feature an autosomal dominant inheritance pattern. Although many of these are rare syndromes, they account for at least 5–10% of all cancer incidences (Refer to Figure 1).
An inherited cancer susceptibility syndrome is usually suspected in families with the following characteristics:
- Two or more relatives with the same type of cancer on the same side of the family
- Several generations affected
- Earlier age of cancer diagnosis than typically seen for that cancer type
- Individuals with multiple primary cancers
- The occurrence of cancers in one family, which are known to be genetically-related (such as breast and ovarian cancer, or colon and uterine cancer) and
- The occurrence of non-malignant conditions and cancer in the same person and/or family (Refer to Figure 2)
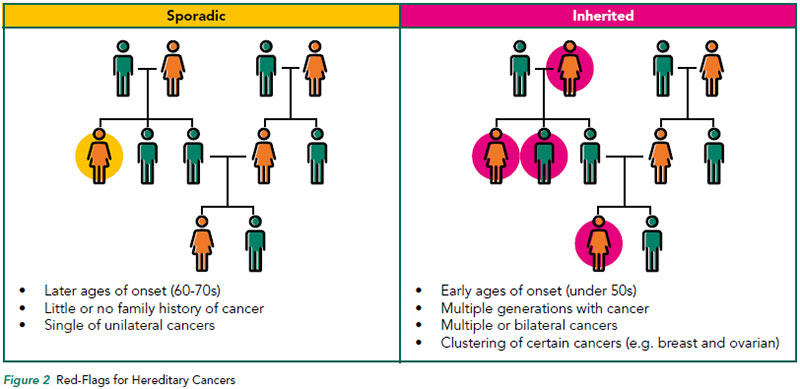
However, due to phenotypic variability, age-related penetrance, and gender-specific cancer risks, many families with an inherited cancer syndrome will not meet these criteria and may be missed by clinicians.
Cancers that arise as a result of a germline predisposition are typically managed differently from those that arise sporadically. Patients may undergo more extensive local therapy if they are at increased risk for metachronous malignancy.
Obvious examples include the consideration of bilateral mastectomy instead of breast conservation in patients with breast cancer carrying a germline BRCA1 or BRCA2 mutation, and subtotal colectomy instead of limited resection in patients with colorectal cancer who have Lynch syndrome. Furthermore, germline mutations have also been shown to be predictive of differential response to treatment approaches.
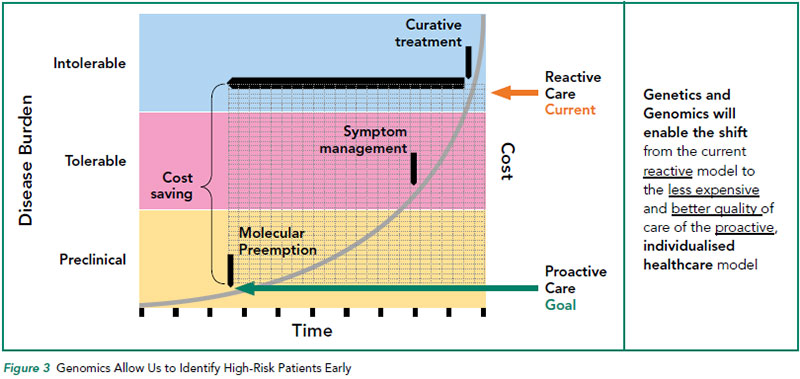
Perhaps the most important application of genetic testing is in individual cancer risk assessment. In addition to enabling successful risk reduction strategies for the at risk patient, identification of germline mutations in cancer-associated genes also informs family members of their individual cancer risks such that appropriate intervention can be offered for affected family members or unwarranted surveillance can be avoided in those unaffected. Knowing who is at high-risk allows us to intensify screening to those who will benefit from the surveillance, resulting in earlier cancer detection and overall cost-savings (Refer to Figure 3).
The Cancer Genetics Service (CGS) at the National Cancer Centre Singapore investigates genetic diseases caused by germ-line (inherited) mutations, and provides comprehensive clinical genetic services, education and support to patients and family members. Our team of clinical cancer geneticists and genetic counsellors work closely with primary care providers and specialists to help incorporate genetic information into a patient’s overall healthcare plan.
Throughout the CGS’s defined four-step genetic testing process, our highly trained and compassionate genetic counsellors help patients navigate the complexities of genetic testing, translating complicated data into information patients can readily understand.
WHEN REFERRAL IS INDICATED
Referrals are made when:
- a patient or family member is diagnosed with cancer or other diseases at a young age
- a patient presents with features of a genetic or inherited condition or with a family history of such a condition, or
- when a patient with a strong family history of a disease or predisposing ethnicity has questions about genetic testing.
Step 1: Risk Assessment
The genetic testing process at the Cancer Genetics Service (CGS) at NCCS begins prior to the first appointment.
Patients provide detailed family health information, focusing on family members that have been diagnosed with genetic or developmental conditions, at what age and if/when family members have died from the disorder(s). The family history allows genetic counsellors to identify patterns of inheritance.
During the initial appointment, the genetic counsellor reviews findings and completes the patient’s medical history.
A physical exam may be performed by the doctor to look for syndromic features such as hand length, arm span, ear length or head circumference (Refer to Picture 4).
Step 2: Consultation
With the risk assessment completed, the
genetic counsellor, often in conjunction with a cancer geneticist, determines whether a genetic testing is advised, and the appropriate test(s) for the patient.
Genetic counsellors provide a great deal of education and counselling prior to and after testing, helping patients understand and consider their options. They explain the basics of genetics and describe the conditions being investigated. If a genetic condition is confirmed, they also walk the patients through the emotional, psychological, and social impact of the genetic results to the patient and family members.
Some patients want to know their risk, even when effective treatments for the condition do not yet exist, while other patients would rather not know in that circumstance.
Genetic counsellors are adept at communicating sensitive genetic information to patients and relatives, taking into account family dynamics and individual coping styles. They also discuss costs and insurance. The decision to pursue genetic testing is personal and genetic counsellors help guide patients and families through the difficulties in decision-making process.
Step 3: Testing
Generally blood is drawn on the same day or at next visit to allow patients time to digest the information provided during consultation. Results may take 2 weeks to 4 months, depending on the extent of testing.
Step 4: Post-Test Genetic Counselling
Gene test results are discussed with the patient by phone or during a follow-up appointment with a genetic counsellor, as part of the standard of care. Next steps may include additional testing, testing of other family members and discussion of gene-informed medical management issues.
If a test result shows a genetic mutation, we carefully discuss future risks and prevention options and formulate an individualised medical management plan based on the specific mutated gene in the context of personal and family history.
For example, if a patient tests positive for a BRCA1/2 breast cancer mutation, we review the statistical risk of developing breast and/or ovarian cancer and their ages of risk; discuss high-risk surveillance (e.g., breast MRI); prevention options, including surgery; and recommend that other family members be screened.
We take time to address patient concerns and answer questions from relatives. We work closely with the patient’s primary physician. Like all investigations, some patients will have results which are not informative or have a result that is of uncertain significance. Our team closely reviews all such results with the clinical laboratory as well as with the patient to tailor management according to their personal and family histories.
If a research opportunity is available, we discuss the risks, benefits and limitations of participation. We also provide ongoing care for patients with genetic conditions, coordinate multidisciplinary medical appointments and help patients access resources related to their condition.
Since 2014, the Cancer Genetics Service at NCCS has seen a sharp rise in demand by patients requesting for genetic testing. Over the last 3 years, more than 1800 patients have had genetic risk assessment and clinical testing.
On average, one-fifth (20%) of the patient tested are positive for one of the cancer predisposition
syndromes.
Our team has studied and published on the local prevalence of inherited genetic mutations in breast cancers, gynaecological cancers, childhood cancers as well as in rare cancers such sarcomas and endocrine-related cancers. Prevalence varies according to cancer type, for example, about 20% of all patients with ovarian cancers and 5% of all patients with colorectal cancers have underlying genetic predisposition.
To understand one’s genetic profile is typically a very personal choice with many factors impacting how patients decide. A personal history and/or family history of cancer may motivate one patient to have testing but may also be the reason why another patient opt against testing.
An in-depth interview study conducted among patients who have undergone testing showed that Singaporean patients are heavily influenced by their primary doctors’ opinions on whether they should proceed with genetic testing. It is therefore very important to elevate genetic literacy amongst clinicians and primary care doctors so that they may identify patients appropriately for a genetics referral.
By: Dr Joanne Ngeow (BMedSci, MBBS, FRCP, MPH) is a Senior Consultant at the Division of Medical Oncology at the National Cancer Centre Singapore, and Associate Professor (Genomic Medicine) at the Lee Kong Chian School of Medicine, Nanyang Technological University Singapore.
Dr Ngeow currently heads the Cancer Genetics Service at the National Cancer Centre Singapore, with an interest in caring for families with hereditary cancer syndromes. She was awarded consecutive fellowships by the National Medical Research Council and the Ambrose Monell Foundation to complete formal clinical and wet bench training in Cancer Genomic Medicine at the Genomic Medicine Institute, Cleveland Clinic, Ohio. Dr Ngeow is funded by the National Medical Research Council and Ministry of Health to explore gene-environment interactions in cancer predispose to cancer initiation and progression as well as the implementation of genomics into routine clinical care.
GPs can call for appointments through the GP Appointment Hotline at 6436 8288 for more information.
